 Compare the different ways to write the reactions. Note the way precipitation reactions appear as molecular equations and net ionic equations. A solid formed by a composition reaction. After youre done simulating a rainstorm or a snowstorm, scroll down to learn more about. Learning goal 1: Identify the statement that best describes a precipitate. In conclusion, Precipitate reactions are an essential part of chemistry and have numerous applications in various fields. Here are common examples of precipitation reactions. Make it rain and snow in this precipitation simulator Set the air temperature (green) and dew point (yellow) in the four different altitudes and see what type of precipitation will fall to the ground. A sample of the unknown substance is mixed with a known substance, and if a precipitate forms, the unknown substance is identified. In forensic science, precipitate reactions are used to identify unknown substances. Precipitates form when the ions experience an electrostatic attraction to each other and they stick together, forming crystals. In some cases, a medication can be isolated and purified through precipitation. Precipitate reactions are used in the production of medications. 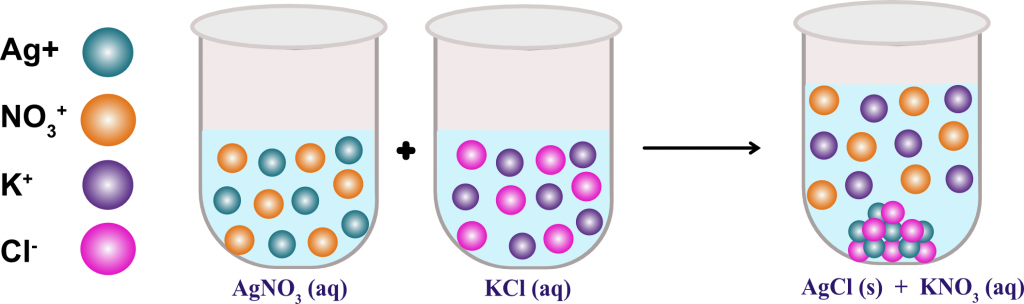
For example, adding Aluminum Sulfate to water causes impurities to precipitate out of the water. In water treatment, precipitate reactions are used to remove impurities from water. If this new compound is insoluble in water, it will precipitate out of the solution. When the ions of the two solutions combine, they form a new compound. When two aqueous solutions are mixed, the ionic compounds present in each solution break down into their respective ions. Precipitation Reactions Experiment: This virtual lab focuses on observing and recording data from several precipitation (double replacement reactions). This can occur when solutions containing ionic compounds. Precipitation reactions occur when two aqueous solutions combine and one of the products is insoluble in water. A precipitate is a solid formed in a chemical reaction that is different from either of the reactants. The process that forms a precipitate is called precipitation. When Calcium Chloride reacts with Sodium Carbonate, it produces Calcium Carbonate precipitate and Sodium Chloride.ĬaCl2 (aq) + Na2CO3 (aq) → CaCO3 (s) + 2NaCl (aq) A precipitate is an insoluble compound that is formed in a liquid solution by mixing two soluble salts together. 
Pb(NO3)2 (aq) + 2KI (aq) → PbI2 (s) + 2KNO3 (aq)ģ. Free Essay: Conclusion: The the precipitation reactions lab eight out of the fourteen occurring reactions were precipitates. When Lead Nitrate reacts with Potassium Iodide, it produces Lead Iodide precipitate and Potassium Nitrate. When Silver Nitrate reacts with Sodium Chloride, it produces Silver Chloride precipitate and Sodium Nitrate.ĪgNO3 (aq) + NaCl (aq) → AgCl (s) + NaNO3 (aq) Precipitation reactions are common in chemistry and are used to isolate, purify and identify various compounds. 
The solid formed is called the precipitate. A precipitate reaction is a chemical reaction in which two aqueous solutions combine to form an insoluble solid. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
